Iron is a mineral used in the human body that is essential for good health. It is produced via the bombardment of lead-208 atoms with iron-58 atoms. Hassium is extremely radioactive, and as such is not found naturally in the Earth's crust. It can also be obtained as a waste product from refining nickel. 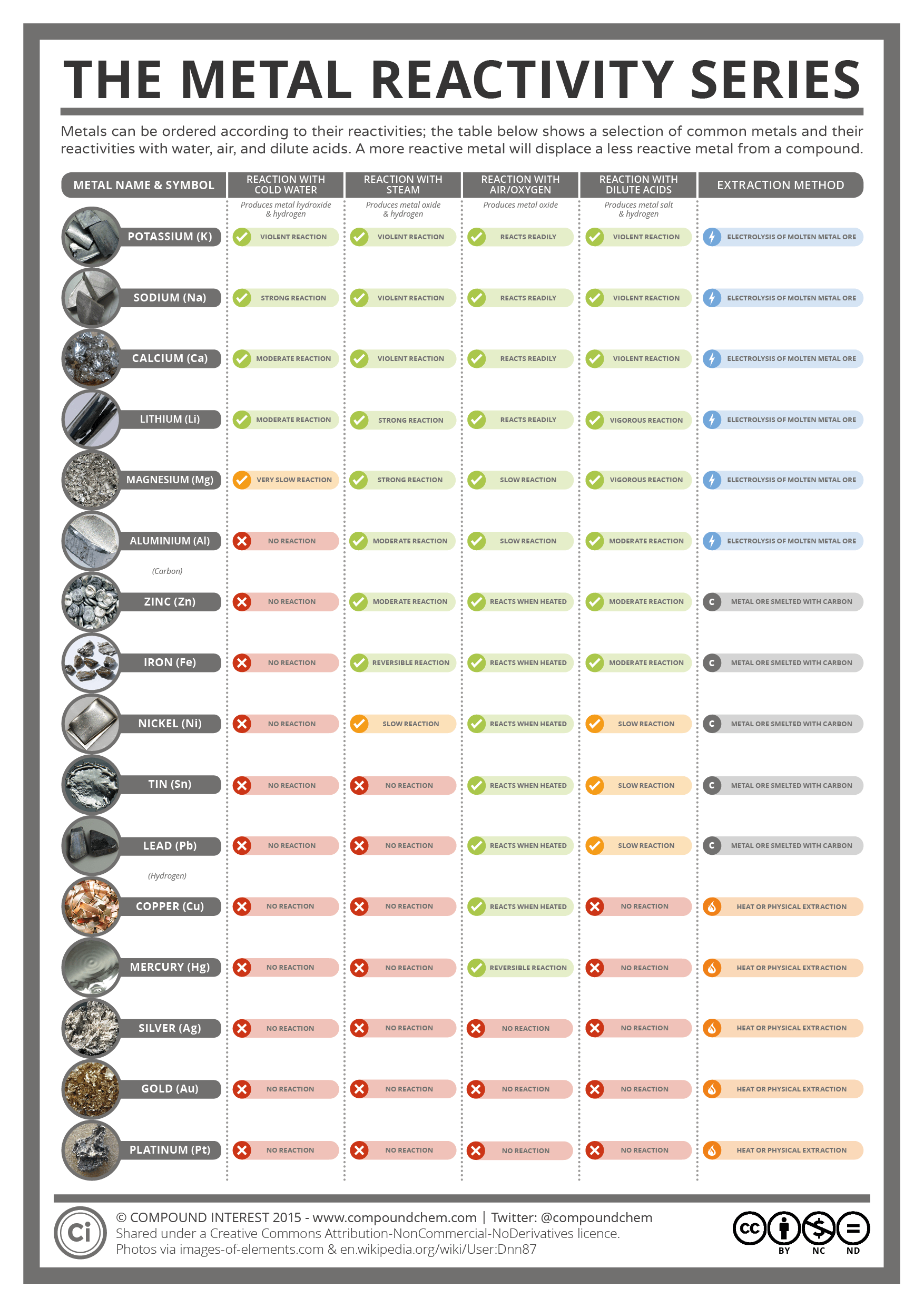
It can be commercially obtained as a waste product from refining nickel. It is often found in minerals such as pentlandite and pyroxinite. Ruthenium is a very rare metal in Earth's crust. Iron is commercially produced by heating these minerals in a blast furnace with coke and calcium carbonate. It is found in many minerals, such as hematite, magnetite, and taconite. In terms of mass, iron is the fourth most common element within the Earth's crust. Though despite its radioactivity, chemists have formed hassium tetroxide and sodium hassate(VII) through various means. A few singular chemistry-related properties have been measured, such as enthalpy of adsorption of hassium tetroxide, but properties of hassium metal remain unknown and only predictions are available. Very few properties of hassium or its compounds have been measured this is due to its extremely limited and expensive production and the fact that hassium (and its parents) decays very quickly. Because of its hardness, brittleness, low vapor pressure (the lowest of the platinum-group metals), and very high melting point (the fourth highest of all elements, after carbon, tungsten, and rhenium), solid osmium is difficult to machine, form, or work. 
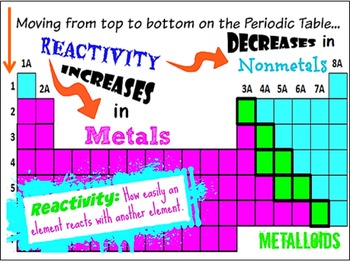
The hardness of osmium is moderately high at 4 GPa. Correspondingly, its bulk modulus is extremely high, reported between 395 and 462 GPa, which rivals that of diamond (443 GPa). Osmium is a hard but brittle metal that remains lustrous even at high temperatures. The ruthenium plate is applied to the electrical contact and electrode base metal by electroplating or sputtering. With its similar properties to and lower cost than rhodium, electric contacts are a major use of ruthenium. electrolytic iron) are more resistant to corrosion.īecause it hardens platinum and palladium alloys, ruthenium is used in electrical contacts, where a thin film is sufficient to achieve the desired durability. Unlike the oxides of some other metals that form passivating layers, rust occupies more volume than the metal and thus flakes off, exposing more fresh surfaces for corrosion. Iron reacts readily with oxygen and water to produce brown-to-black hydrated iron oxides, commonly known as rust. Pristine and smooth pure iron surfaces are a mirror-like silvery-gray. The following is copied from the pages of Iron, Ruthenium, Osmium, and Hassium respectively. Like other groups, the members of this family show patterns in electron configuration, especially in the outermost shells, resulting in trends in chemical behavior. While groups (columns) of the periodic table are usually named after their lightest member (as in "the oxygen group" for group 16), iron group has historically been used differently most often, it means a set of adjacent elements on period (row) 4 of the table that includes iron, such as chromium, manganese, iron, cobalt, and nickel, or only the last three, or some other set, depending on the context. The elements in this group are all transition metals that lie in the d-block of the periodic table. system", or "VIII" in the old IUPAC (pre-1990) "European system" (and in Mendeleev's original table). In the older group naming systems, this group was combined with groups 9 and 10 and called group "VIIIB" in the Chemical Abstracts Service (CAS) "U.S. It should not be confused with "group VIIIA" in the CAS system, which is group 18 (current IUPAC), the noble gases. "Group 8" is the modern standard designation for this group, adopted by the IUPAC in 1990. It consists of iron (Fe), ruthenium (Ru), osmium (Os) and hassium (Hs). Group 8 is a group (column) of chemical elements in the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
